4.2 Reduction to two dimensions
A system of four differential equations, such as the Hodgkin-Huxley model, is difficult to analyze, so that normally we are limited to numerical simulations. A mathematical analysis is, however, possible for a system of two differential equations.
In this section we perform a systematic reduction of the four-dimensional Hodgkin-Huxley model to two dimensions. To do so, we have to eliminate two of the four variables. The essential ideas of the reduction can also be applied to detailed neuron models that may contain many different ion channels. In these cases, more than two variables would have to be eliminated, but the procedure would be completely analogous (258).
| A | B |
|---|---|
|
 |
| A | B |
|---|---|
|
 |
4.2.1 General approach
We focus on the Hodgkin-Huxley model discussed in Chapter 2 and start with two qualitative observations. First, we see from Fig. 2.3B that the time scale of the dynamics of the gating variable is much faster than that of the variables and . Moreover, the time scale of is fast compared to the membrane time constant of a passive membrane, which characterizes the evolution of the voltage when all channels are closed. The relatively rapid time scale of suggests that we may treat as an instantaneous variable. The variable in the ion current equation (2.5) of the Hodgkin-Huxley model can therefore be replaced by its steady-state value, . This is what we call a quasi steady state approximation which is possible because of the ’separation of time scales’ between fast and slow variables.
Second, we see from Fig. 2.3B that the time constants and have similar dynamics over the voltage . Moreover, the graphs of and in Fig. 2.3A are also similar. This suggests that we may approximate the two variables and by a single effective variable . To keep the formalism slightly more general we use a linear approximation with some constants and set . With , , and , equations (2.4) - (2.5) become
| (4.3) |
or
| (4.4) |
with , and some function . We now turn to the three equations (2.2.2). The equation has disappeared since is treated as instantaneous. Instead of the two equations (2.2.2) for and , we are left with a single effective equation
| (4.5) |
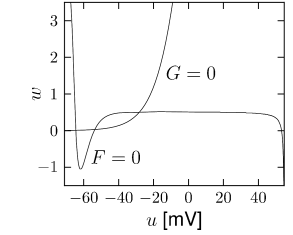
where is a parameter and a function that interpolates between and (see Section 4.2.2). Eqs. (4.4) and (4.5) define a general two-dimensional neuron model. If we start with the Hodgkin-Huxley model and implement the above reduction steps we arrive at functions and which are illustrated in Figs. 4.3A and 4.4A. The mathematical details of the reduction of the four-dimensional Hodgkin-Huxley model to the two equations (4.4) and (4.5) are given below.
Before we go through the mathematical steps, we present two examples of two-dimensional neuron dynamics which are not directly derived from the Hodgkin-Huxley model, but are attractive because of their mathematical simplicity. We will return to these examples repeatedly throughout this chapter.
Example: Morris-Lecar model
Morris and Lecar ( 352 ) proposed a simplified two-dimensional description of neuronal spike dynamics. A first equation describes the evolution of the membrane potential , the second equation the evolution of a slow ‘recovery’ variable . The Morris-Lecar equations read
| (4.6) | |||||
| (4.7) |
If we compare Eq. (4.6) with Eq. (4.3), we note that the first current term on the right-hand side of Eq. (4.3) has a factor which closes the channel for high voltage and which is absent in (4.6). Another difference is that neither nor in Eq. (4.6) have exponents. To clarify the relation between the two models, we could set and . In the following we consider Eqs. (4.6) and (4.7) as a model in its own right and drop the hats over and .
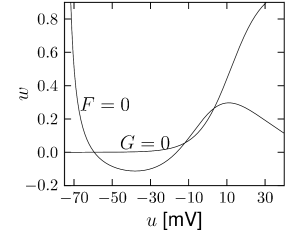
The equilibrium functions shown in Fig. 2.3A typically have a sigmoidal shape. It is reasonable to approximate the voltage dependence by
| (4.8) | |||||
| (4.9) |
with parameters , and to approximate the time constant by
| (4.10) |
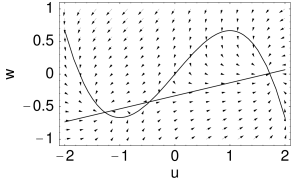
with a further parameter . With the above assumptions, the zero-crossings of functions and of the Morris-Lecar model have the shape illustrated in Fig. 4.3B.
The Morris-Lecar model (4.6)–(4.10) gives a phenomenological description of action potentials. We will see later on that the mathematical conditions for the firing of action potentials in the Morris-Lecar model can be discussed by phase plane analysis.
Example: FitzHugh-Nagumo model
FitzHugh and Nagumo were probably the first to propose that, for a discussion of action potential generation, the four equations of Hodgkin and Huxley can be replaced by two, i.e., Eqs. (4.4) and (4.5). They obtained sharp pulse-like oscillations reminiscent of trains of action potentials by defining the functions and as
| (4.11) |
where is the membrane voltage and is a recovery variable (152; 356). Note that both and are linear in ; the sole non-linearity is the cubic term in . The FitzHugh-Nagumo model is one of the simplest model with non-trivial behavior lending itself to a phase plane analysis, which will be discussed below in Sections 4.3 – 4.5.
4.2.2 Mathematical steps (*)
| A | B |
|---|---|
|
 |
The reduction of the Hodgkin-Huxley model to Eqs. (4.4) and (4.5) presented in this paragraph is inspired by the geometrical treatment of Rinzel (439); see also the slightly more general method of Abbott and Kepler (2) and Kepler et al. (258).
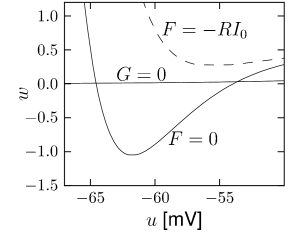
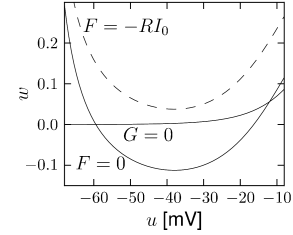
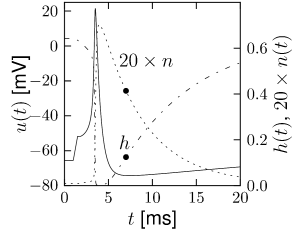
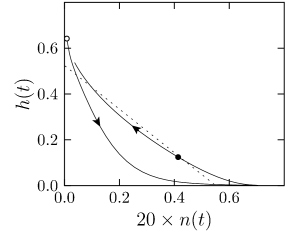
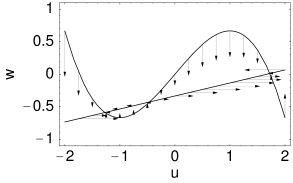
The overall aim of the approach is to replace the variables and in the Hodgkin-Huxley model by a single effective variable . At each moment of time, the values can be visualized as points in the two-dimensional plane spanned by and ; cf. Fig. 4.5B. We have argued above that the time course of the scaled variable is expected to be similar to that of . If, at each time, were equal to , then all possible points would lie on the straight line which changes through and . It would be unreasonable to expect that all points that occur during the temporal evolution of the Hodgkin-Huxley model fall exactly on that line. Indeed, during an action potential (Fig. 4.5A), the variables and stay close to a straight line, but are not perfectly on it (Fig. 4.5B). The reduction of the number of variables is achieved by a projection of those points onto the line. The position along the line gives the new variable ; cf. Fig. 4.6. The projection is the essential approximation during the reduction.
To perform the projection, we will proceed in three steps. A minimal condition for the projection is that the approximation introduces no error while the neuron is at rest. As a first step, we therefore shift the origin of the coordinate system to the rest state and introduce new variables
| (4.12) | |||||
| (4.13) |
At rest, we have .
Second, we turn the coordinate system by an angle which is determined as follows. For a given constant voltage , the dynamics of the gating variables and approaches the equilibrium values . The points as a function of define a curve in the two-dimensional plane. The slope of the curve at yields the rotation angle via
| (4.14) |
Rotating the coordinate system by turns the abscissa of the new coordinate system in a direction tangential to the curve. The coordinates in the new system are
| (4.15) |
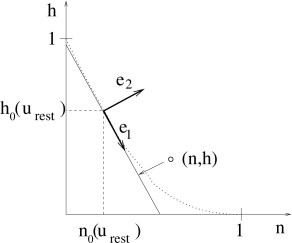
Third, we set and retain only the coordinate along . The inverse transform,
| (4.16) |
yields and since . Hence, after the projection, the new values of the variables and are
| (4.17) | |||||
| (4.18) |
In principle, can directly be used as the new effective variable. From (4.15) we find the differential equation
| (4.19) |
We use Eq. (2.6) and replace, on the right-hand side, and by (4.17) and (4.18). The result is
| (4.20) |
which is of the form , as desired.
To see the relation to Eqs. (4.3) and (4.5), it is convenient to rescale and define
| (4.21) |
If we introduce and , we find from Eq. (4.17) the variable and from Eq. (4.18) , which are exactly the approximations that we have used in (4.3). The differential equation for the variable is of the desired form and can be found from Eq. (4.20) and (4.21). The resulting function of the two-dimensional model is illustrated in Fig. 4.3A.
Example: Further simplification
We may further approximate the time constants and by a common function so that the dynamics of is
| (4.22) |
with a new equilibrium function that is a linear combination of the functions and . From Eqs. (4.20) and (4.21) we find
| (4.23) |
In practice, and can be fitted by the expressions (4.9) and (4.10), respectively.
| A | B |
|---|---|
|
 |